FDA clears boosters for Moderna and J&J COVID-19 vaccines
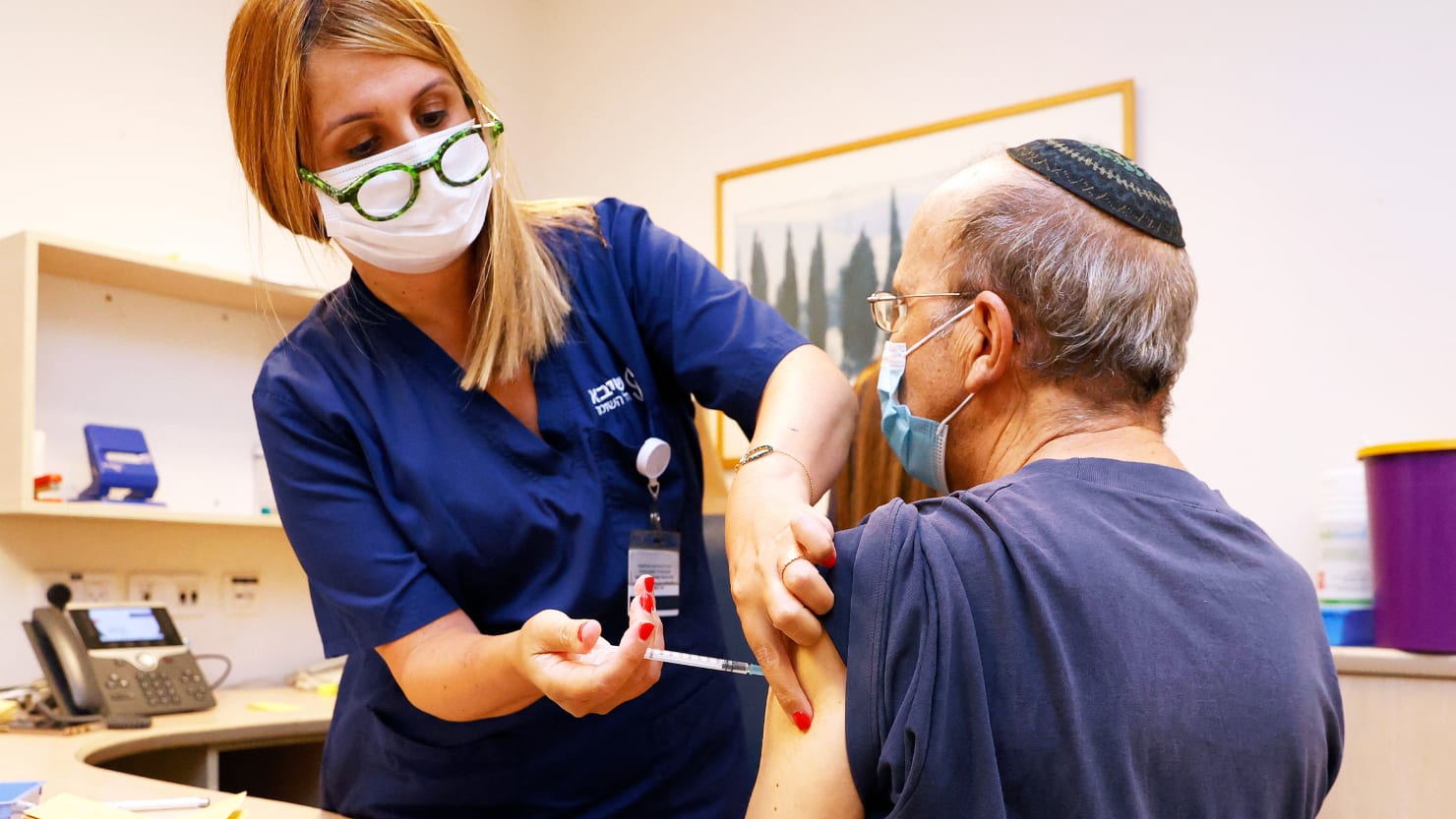
The Food and Drug Administration authorized booster doses for the Moderna and Johnson & Johnson COVID-19 vaccines, following its authorization of a third Pfizer / BioNTech shot in September.
The agency also said today that it will allow “mix-and-match” boosters — eligible people who are already fully vaccinated can get a booster dose of any of the three vaccines, regardless of which they started with.
As with the Pfizer / BioNTech booster, the FDA cleared third Moderna shots for vulnerable groups, including people 65 years of age and older, those who are at high risk of severe disease, and people at high risk of exposure to COVID-19 at work.
The booster for the Moderna vaccine, though, will be a half-dose. Both vaccines are mRNA vaccines that contain tiny snippets of the coronavirus’ genetic material. But the Moderna vaccine has a larger dose: it’s 100 micrograms, compared with the 30 micrograms in the Pfizer / BioNTech shot. The company says a third, 50 microgram half-dose of the Moderna vaccine could have fewer side effects while still giving strong protection.
Research shows that the Moderna shots have more durable protection against infection from COVID-19 than the Pfizer / BioNTech shots — they stay more effective months after the initial two-shot series, while the efficacy for the Pfizer / BioNTech shots appears to drop off. Both maintain strong protection against hospitalization and death.



/cloudfront-us-east-2.images.arcpublishing.com/reuters/WH6YZXYGUVPP3EGR3KFXLDHZ2Q.jpg)