Solid-state battery - Wikipedia
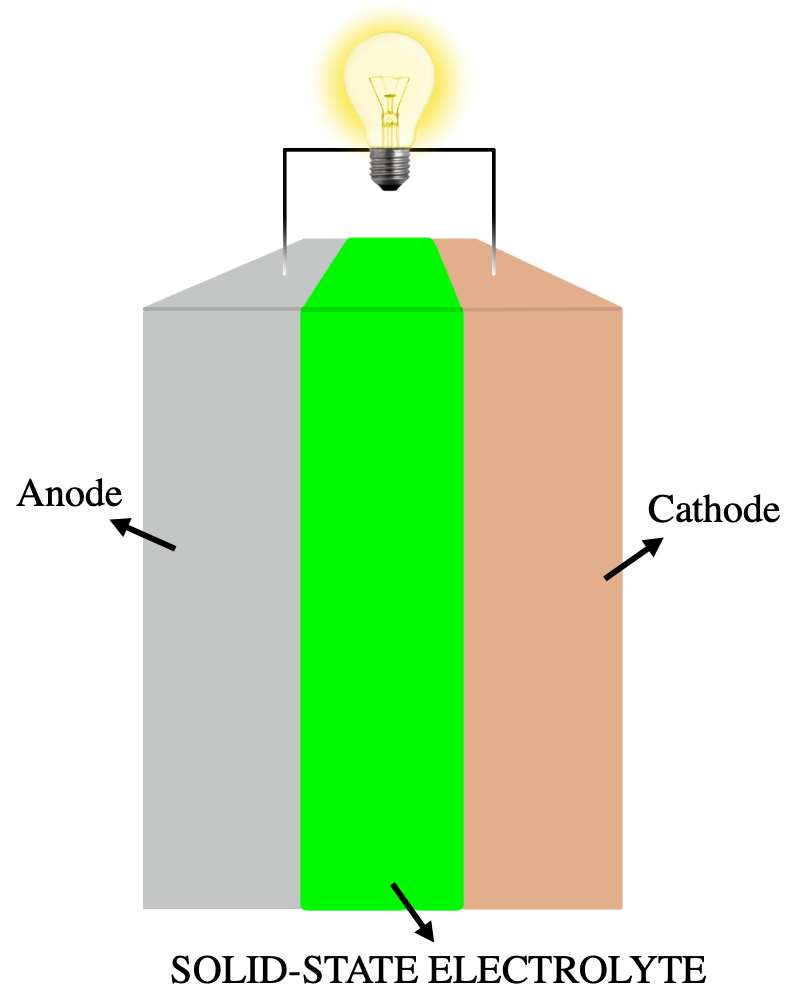
A solid-state battery is an electrical battery that uses a solid electrolyte for ionic conductions between the electrodes, instead of the liquid or gel polymer electrolytes found in conventional batteries.[ 1] Solid-state batteries theoretically offer much higher energy density than the typical lithium-ion or lithium polymer batteries.[ 2]
While solid electrolytes were first discovered in the 19th century, several problems prevented widespread application. Developments in the late 20th and early 21st century generated renewed interest in the technology, especially in the context of electric vehicles.
Solid-state batteries can use metallic lithium for the anode and oxides or sulfides for the cathode, increasing energy density. The solid electrolyte acts as an ideal separator that allows only lithium ions to pass through. For that reason, solid-state batteries can potentially solve many problems of currently used liquid electrolyte Li-ion batteries, such as flammability, limited voltage, unstable solid-electrolyte interface formation, poor cycling performance, and strength.[ 5]
Materials proposed for use as electrolytes include ceramics (e.g., oxides, sulfides, phosphates), and solid polymers. Solid-state batteries are found in pacemakers, and in RFID and wearable devices[citation needed ]. Solid-state batteries are potentially safer, with higher energy densities. Challenges to widespread adoption include energy and power density, durability, material costs, sensitivity, and stability.[ 6]